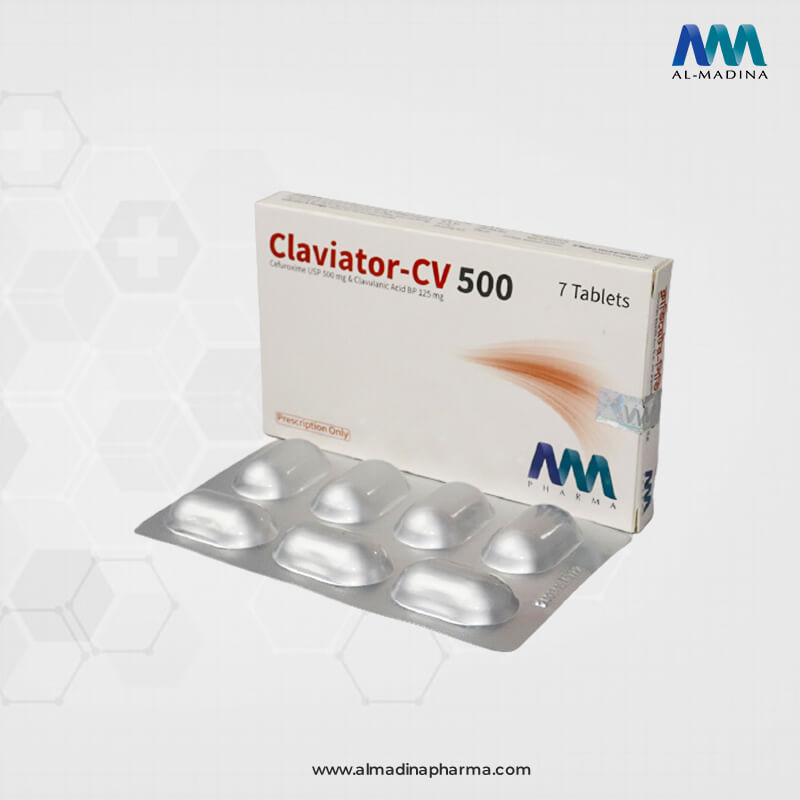
Claviator-CV
৳ 250 – ৳ 420
Brand Name: Claviator-CV
Generic Name: Cefuroxime Axetil USP & Potassium Clavulanate BP
Therapeutic Class: Antibiotic
- Additional information
- Presentation
- Description
- Indication
- Dose And Administration
- Side Effects
- Warning & Precaution
- Contraindication
- Use In Pregnancy And Lactation
- Packaging
- Drug Interaction
- Overdose
- Reviews (0)
Additional information
| STRENGTH | 70ml, 250mg, 500mg |
|---|
Presentation
CLAVIATOR-CV 250 Tablet: Each film coated tablet contains Cefuroxime Axetil USP 300.01 mg equivalent to Cefuroxime 250.00 mg and diluted Potassium Clavulanate BP 74.40 mg equivalent to Clavulanic Acid 62.50 mg
CLAVIATOR-CV 500 Tablet: Each film coated tablet contains Cefuroxime Axetil USP 600.02 mg equivalent to Cefuroxime 500.00 mg and diluted Potassium Clavulanate BP 148.80 mg equivalent to Clavulanic Acid 125.00 mg.
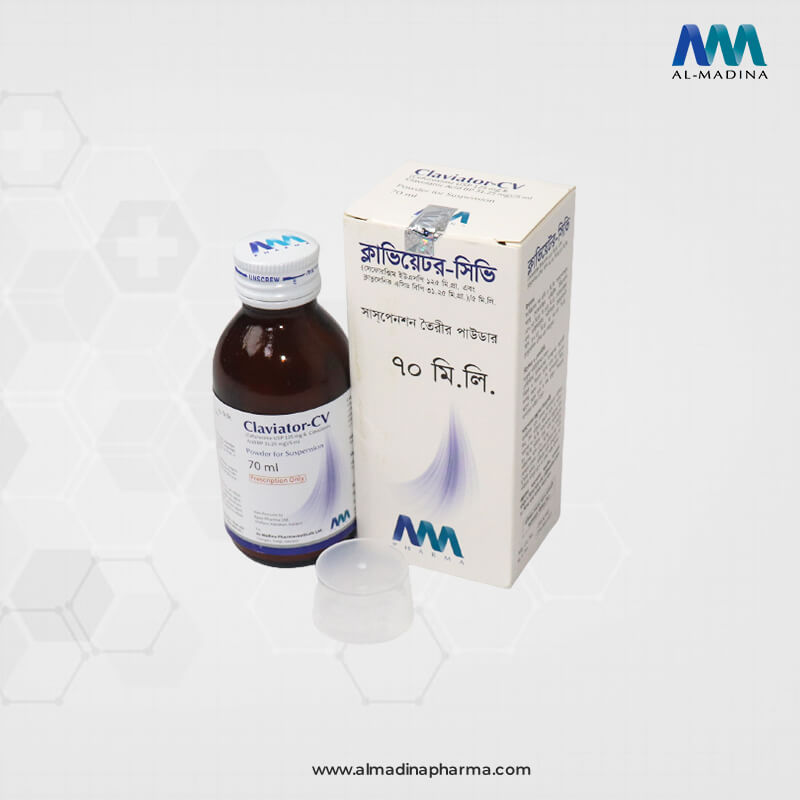
CLAVIATOR-CV 70 ml Powder for Suspension: After reconstitution, each 5 ml suspension contains Cefuroxime Axetil USP 150.00 mg equivalent to Cefuroxime 125.00 mg and diluted Potassium Clavulanate BP 37. 25 mg as Clavulanic Acid 31.25 mg.
Description
Cefuroxime is Second generation Cephalosporin. Cefuroxime is a well-characterized and effective antibacterial agent, which has broad-spectrum bactericidal activity against a wide range of common pathogens, including B-lactamase producing strains. Cefuroxime has good stability to bacterial ß-lactamase and consequently, is active against many ampicillin-resistant and amoxycillin-resistant strains. Clavulanic acid has a similar structure to the beta-lactam antibiotics but binds irreversibly to the beta-lactamase enzymes. The presence of clavulanic acid in CLAVIATOR-CV formulations protects cefuroxime from degradation by beta-lactamase enzymes and effectively extends the antibacterial spectrum of cefuroxime to include many bacteria normally resistant to cefuroxime and other cephalosporins.
Indication
• Pharyngitis/Tonsillitis caused by Streptococcus pyogenes.
• Acute Bacterial Otitis Media caused by Streptococcus pneumoniae, Haemophilus influenzae (including beta- lactamase producing strains), Moraxella catarrhalis (including beta-lactamase producing strains) or Streptococcus pyogenes.
• Acute Bacterial Maxillary Sinusitis caused by Streptococcus pneumoniae or Haemophilus influenzae.
• Acute Bacterial Exacerbations of Chronic Bronchitis and Secondary Bacterial Infections of Acute Bronchitis caused by Streptococcus pneumoniae, Haemophilus influenzae or Haemophilus parainfluenzae.
• Uncomplicated Skin and Skin-Structure Infections caused by Staphylococcus aureus (including beta-lactamase producing strains) or Streptococcus pyogenes.
• Uncomplicated Urinary Tract Infections caused by Escherichia coli or Klebsiella pneumoniae.
• Uncomplicated Gonorrhea (urethral and endocervical) caused by Neisseria gonorrhoeae and Uncomplicated Gonorrhea rectal in females caused by non-penicillinase producing strains of Neisseria gonorrhoeae.
• Early Lyme disease (erythema migrans) caused by Borrelia burgdorferi.
• Septicemia caused Staphylococcus aureus. Streptococcus pneumoniae. Escherichiacoli. Haemophilus influenzae (including ampicillin-resistant strains) and Klebsiella spp. • Meningitis caused by Streptococcus pneumoniae, Haemophilus influenzae (including ampicillin-resistant strains), Neisseria meningitides and Staphylococcus aureus (penicil linase and non-penicillinase producing strains).
• Switch therapy (injectable to oral).
Dose And Administration
CLAVIATOR-CV Tablets: The usual course of therapy with CLAVIATOR-CV tablets is 5 to 7 days for treatment of bronchitis and 7 to 10 days for most other infection.
|
Population/Infection |
Dosage |
Duration(Days) |
|
Adolescents and Adults ( 13 years and Older) |
||
| Pharyngitis / tonsillitis
|
250 mg twice daily
|
5-10 |
| Acute bacterial maxillary sinusitis
|
250 mg twice daily |
10 |
| Acute bacterial exacerbations of chronic bronchitis | 250 or 500 mg twice daily
|
10 |
| Secondary bacterial infections of acute bronchitis | 250 or 500 mg twice daily
|
5-10 |
| Uncomplicated skin and skin- structure infection | 250 or 500 mg twice daily
|
10 |
|
Uncomplicated urinary tract infection
|
250 mg twice daily |
7-10 |
| Uncomplicated gonorrhea
|
1000 mg as single dose |
Single Dose |
|
Early Lyme disease
|
500 mg twice daily
|
20 |
Geriatric Use: The geriatric patients reported somewhat fewer gastrointestinal events and less frequent vaginal candidiasis compared with patients aged 12 to 64 years old; however, no clinically significant differences were reported between the elderly and younger adult patients. Other reported clinical experience has not identified differences in responses between the elderly and younger adult patients.
Side Effects
a) Common: Generally Cefuroxime and Clavulanic acid are well tolerated. However, a few side effects like nausea, vomiting, diarrhea, abdominal discomfort or pain may occur. As with other broad-spectrum antibiotics, prolonged administration of Cefuroxime and Clavulanic acid combination may result in overgrowth of non-susceptible microorganisms.
b) Rare: Rarely (<0.2%) renal dysfunction, anaphylaxis, angioedema, pruritis, rash and serum sickness like urticaria may appear
Warning & Precaution
CLAVIATOR-CV should be given with care to patients receiving treatment with potent diuretics & who have history of colitis.
Contraindication
Patients with known allergy to cephalosporin’s & pseudomembranous colitis are contraindicated.
Use In Pregnancy And Lactation
During pregnancy: All antibiotics should be avoided in the first trimester, if possible. However, CLAVIATOR-CV can be safely used in later pregnancy to treat urinary and other infections.
Packaging
CLAVIATOR-CV 250 Tablet: Each box contains 2×7’s Alu-Alu blister of tablets.
CLAVIATOR-CV 500 Tablet: Each box contains 1×7’s Alu-Alu blister of tablets.
CLAVIATOR-CV 70 ml Powder for Suspension: Amber glass bottle contains powder to prepare 70 ml suspension and a measuring cup.
Drug Interaction
a) With medicine: Concomitant administration of probenecid with CLAVIATOR-CV increases the area under the serum concentration versus time curves by 50%. Drugs that reduces gastric acidity may result in a lower bioavailability of cefuroxime and tend to cancel the effect of postprandial absorption.
b) With Food: Alcohol has no directed effect on CLAVIATOR-CV. However it may aggravate infection / CLAVIATOR-CV induced stomach upset or headache
Overdose
Overdose of CLAVIATOR-CV can cause cerebral irritation leading to convulsions. Serum levels of CLAVIATOR-CV can be reduced by hemodialysis and peritoneal dialysis.

























Reviews
There are no reviews yet.