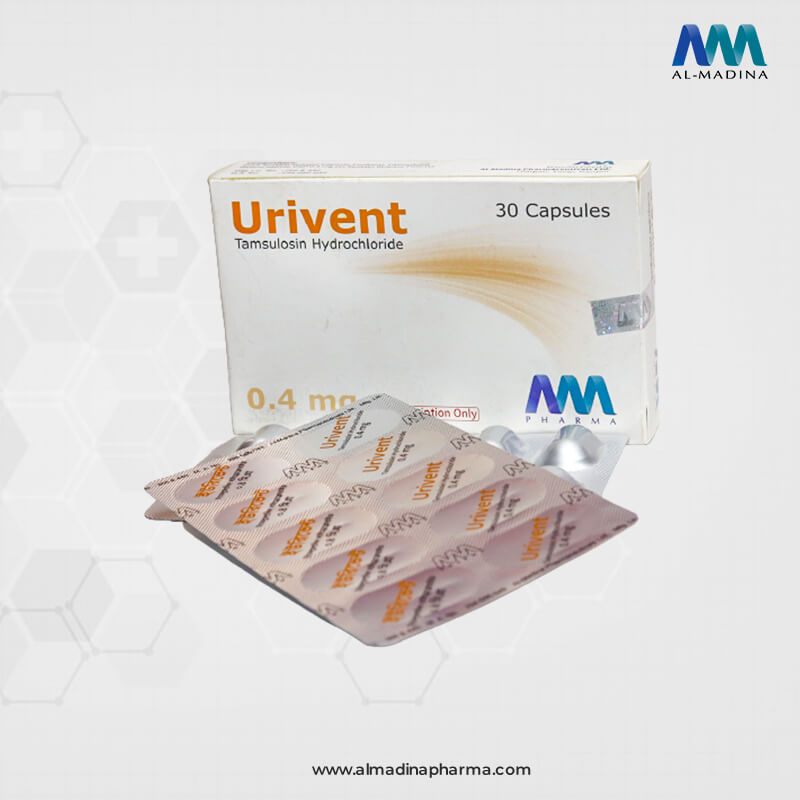
Urivent
৳ 270
Brand Name: Urivent
Generic Name: Tamsulosin Hydrochloride USP
Therapeutic Class: Urinary Alkalizing Agent/Urinary Retention
- Additional information
- Presentation
- Description
- Indication
- Dose And Administration
- Side Effects
- Warning & Precaution
- Contraindication
- Use In Pregnancy And Lactation
- Packaging
- Drug Interaction
- Overdose
- Reviews (0)
Additional information
| STRENGTH | 0.4mg |
|---|
Presentation
Urivent Capsule: Each sustained release capsule contains Tamsulosin Hydrochloride USP 0.4 mg as (sustained release pellets)
Description
Tamsulosin a selective alpha, adrenoceptor blocking agent, exhibits its selectivity for alphaA, adrenoceptors in human prostate. Blockade of these adrenoceptors can cause smooth muscle in the bladder neck and prostate to relax, resulting in an improvement in urine flow rate and a reduction in symptoms of BPH. Absorption of Tamsulosin Hydrochloride capsules 0.4 mg is essentially complete (90%) following oral administration under fasting condition. The time to maximum concentration (Tmax) is reached by four to five hours under fasting conditions and by six seven hours when administered with food. Tamsulosin Hydrochloride is extremely bound to human plasma protein (94% to 99%). Tamsulosin Hydrochoride is extensively metabolized by cytochrome P 450 enzymes in the liver and less than 10% of the dose is excreted in urine as unchanged form. Following intravenous or oral administration of an immediate-release formulation the elimination half-life of Tamnsulosin Hydrochloride in plasma rages form five to seven hours. Because of the absorption rate controlled pharmacokinetics with capsules, the apparent half-life of the Tamsulosin Hydrochloride is approximately 9 to 13 hours in healthy volunteers and 14 to 15 hours in the target population.
Indication
Urivent Capsule is indicated for the treatment of the signs and symptoms of Benign Prostatic Hyperplasia (BPH).
Dose And Administration
The recommended dose of Urivent Capsule is 0.4 mg once daily. It should be administered approximately one half/hour following the same meal each day. For those patients who fail to respond to the 0.4 mg dose after two to four weeks of dosing, the dose of Urivent Capsule can be increased to 0.8 mg once daily. If Urivent Capsule administration is discontinued or interrupted for several days at either the 0.4 mg or 0.8 mg dose, therapy should be started again with the 0.4 mg once daily dose.
Side Effects
a) Common: The following adverse reactions have been reported during the use of Tamsulosin: Dizziness, abnormal ejaculation, and less frequently (1-2%) headache, asthenia, postural hypotension, palpitations, and rhinitis. Gastrointestinal reactions such as nausea, vomiting, diarrhea, and constipation can occasionally occur. Hypersensitivity reactions such as rash, pruritus, and urticaria can occur occasionally. As with other alpha-blockers, drowsiness, blurred vision, dry mouth, or oedema can occur.
b) Rare: Syncope has been reported rarely, and there have been rare reports of angioedema and priapism.
Warning & Precaution
Not indicated for use in women. Severe renal impairment (creatinine clearance is less than 10 mL/min) should be approached with caution as these patients have not been studied. Caution should be exercised in porphyria or allergic reaction to this or any other medicine.
Contraindication
History of orthostatic hypotension. Severe hepatic impairment. As with other alpha 1 blockers, a reduction in blood pressure can occur in individual cases during treatment with tamsulosin, as a result of which, rarely, synocope can occur. At the first signs of orthostatic hypotension (dizziness, weakness) the patient should sit or lie down until the symptoms have disappeared.
Use In Pregnancy And Lactation
Urivent capsules are not indicated for use in women.
Packaging
Urivent Capsule: Each box contains 3 X 10’s capsules in Alu-Alu blister strips.
Drug Interaction
a) With medicine: Increased plasma concentration with strong CYP3A4 inhibitors (e.g. ketoconazole. Moderate CYP3A4 inhibitors (e.g. erythomycin), strong (e.g. paroxetine) or moderate (e.g. terbinafine) CYP2D6 inhibitors may increase exposure of tamsulosin. Increased plasma concertation with cimetidine. Additive effect with other a-adrenergic blocking agents. Concomitant use with PDE5 inhibitor may lead to may symptomatic hypotension. Decreased plasma concentration with.
b) With food: Taking tamsulosin in fasting condition results in a 30% increase in bioavailability. Use caution when combining tamsulosin with alcohol-containing beverages. Alcohol-containing beverages may increase the effects of tamsulosin, which may lower the blood pressure.
Overdose
As overdose of Tamsulosin Hydrochloride capsules lead to hypotension, support the cardiovascular system is of first importance, restoration of blood pressure and normalization of heart rate may be accomplished by keeping the patient in supine position. If this measure is inadequate, then administration of intravenous fluid should be considered. Measures, such as emesis, can be taken to impede absorption. When large quantities are involved, gastric lavage can be applied and activated charcoal and an osmotic laxative, such as sodium sulphate, can be administered.



















Reviews
There are no reviews yet.